
Overall, the burning of the set of cards adds to the entropy of the universe slightly. Some of the energy is transformed into thermal energy and dispersed into the atmosphere. Burning objects causes the potential energy in the materials to change. The cards made of paper have potential chemical energy. The cards would then catch fire and burn spontaneously. If you decide to burn one set of cards, now you’re talking! The set of cards that you decide to burn would undergo an entropy change.įirst, to burn the cards, you apply activation energy by means of lighting a match to it.
So, what is a good definition for entropy? A good approach is to think in terms of heat or energy redistribution, not randomness. So, a change in entropy has not taken place in any of the three groups of cards regardless of how they are arranged. All three groups still have the same arrangement of molecules more or less and they have not undergone any heat or energy redistribution. One idea that often leads to misconceptions is that “entropy is a measure of disorder.” One tends to think about patterns when the word “disorder” or “randomness” is used.Īs described earlier, how do you measure disorder? Is it by how far molecules are apart from each other? Is it by the arrangement or pattern of molecules, particles, or objects? If you have a pack of cards randomly spread out a table next to a pack of cards neatly stacked, you may say one is more random than the other.īelow, group B & C cards are more randomly placed, but the properties of the cards have not changed. There are quite a few variations for the definition of entropy. It is important to understand what they are.Ĭlick here for more information on Thermodynamic Systems Entropy and disorder

When unusable energy increases, it means entropy also increases.Įnergy transfer is studied in three types of system.The Second Law of Thermodynamics states that once energy is changed from usable form to unusable form, it cannot be recovered.Įntropy is a measure of that unusable energy. As energy is used for productivity – such as growing new cells or repairing tissues, useful energy is converted to unusable energy.
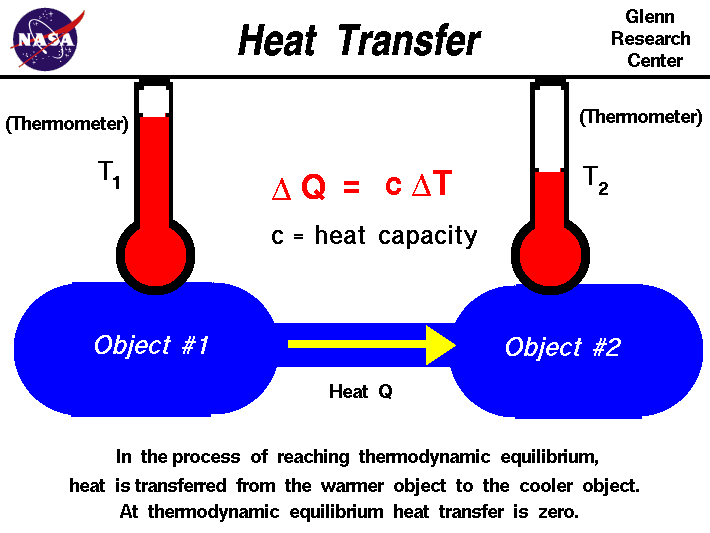
While quantity remains the same (first law), the quality of matter/energy deteriorates. The Second Law of Thermodynamics states that matter/energy gradually deteriorates over time.The First Law of Thermodynamics states that matter/energy cannot be created or destroyed.Thermodynamics is just a branch of physics that studies heat and other forms of energy (mechanical, electrical or chemical) and their relationships between each other. So, disorder is more probable in the universe than order.Įntropy Change from Viziscience on Vimeo. Bricks are not naturally stacked up nicely or become part of a building unless someone has put a lot of effort into making it. Mechanical things tend to wear out and break down. For example, all living things age and die, they don’t get younger. The increase in unusable energy over time is referred to as entropy.įirst of all, entropy is the concept that everything in the universe tends towards disorder.When converting the useful energy to do work, some of the energy is always lost as heat.Solid wood burns to become ash and releases gases.When water flows over a dam, it loses some of its useful energy as heat.What is entropy? Entropy refers to the amount of unusable energy in a system.
